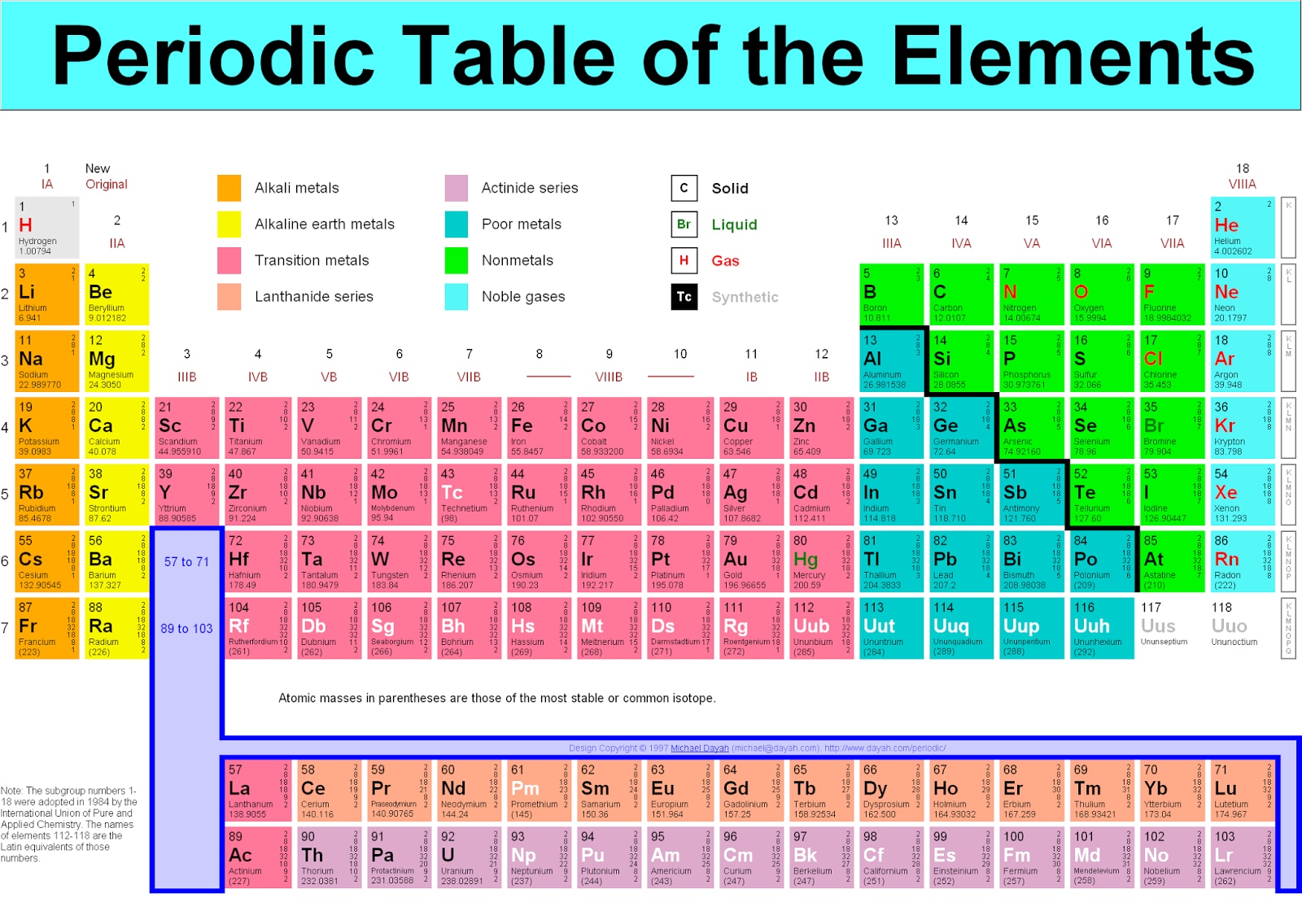
Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. One mole of 'something' contains 6.022 x 1023 entities. While substantially more abundant than gold and silver it importance in history is unmatched and its utility at the macro scale is only matched by its utility at the micro scale. A mole is the unit that measures the amount of a substance. In my opinion youre unlikely to find a metal on the periodic table that has the versatility of copper and still has not been given the respect amongst its peers that it deserves. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. 
The molecular mass is the sum of the masses of the atoms in a molecule. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic mass is the weighted average of the masses of all isotopes of an element. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. The chemical formula for an ethylene monomer is -(CH2-CH2).There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The unity for atomic mass is gram per mol. In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. The lightest chemical element is Hydrogen and the heaviest is Hassium. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. 
Is it ok to simply write 16 down for the mass when calculating the molar mass of a compound containing oxygen (e.g. For example, the mass of oxygen on the periodic table you used for labeling is 16.00.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
